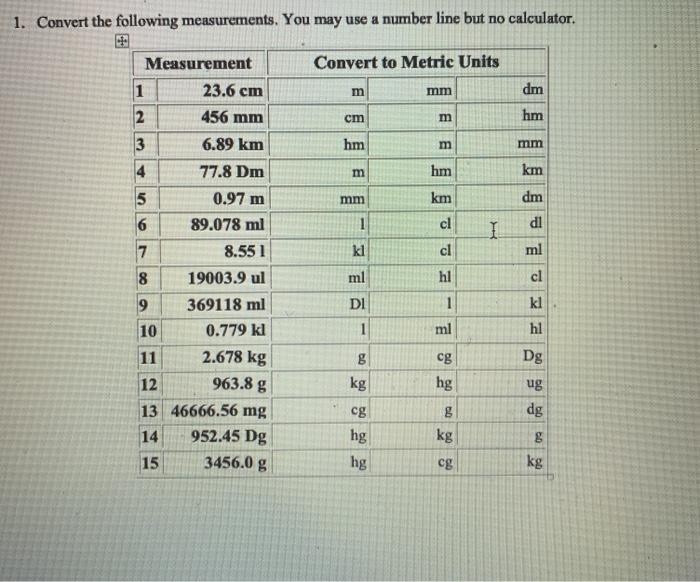
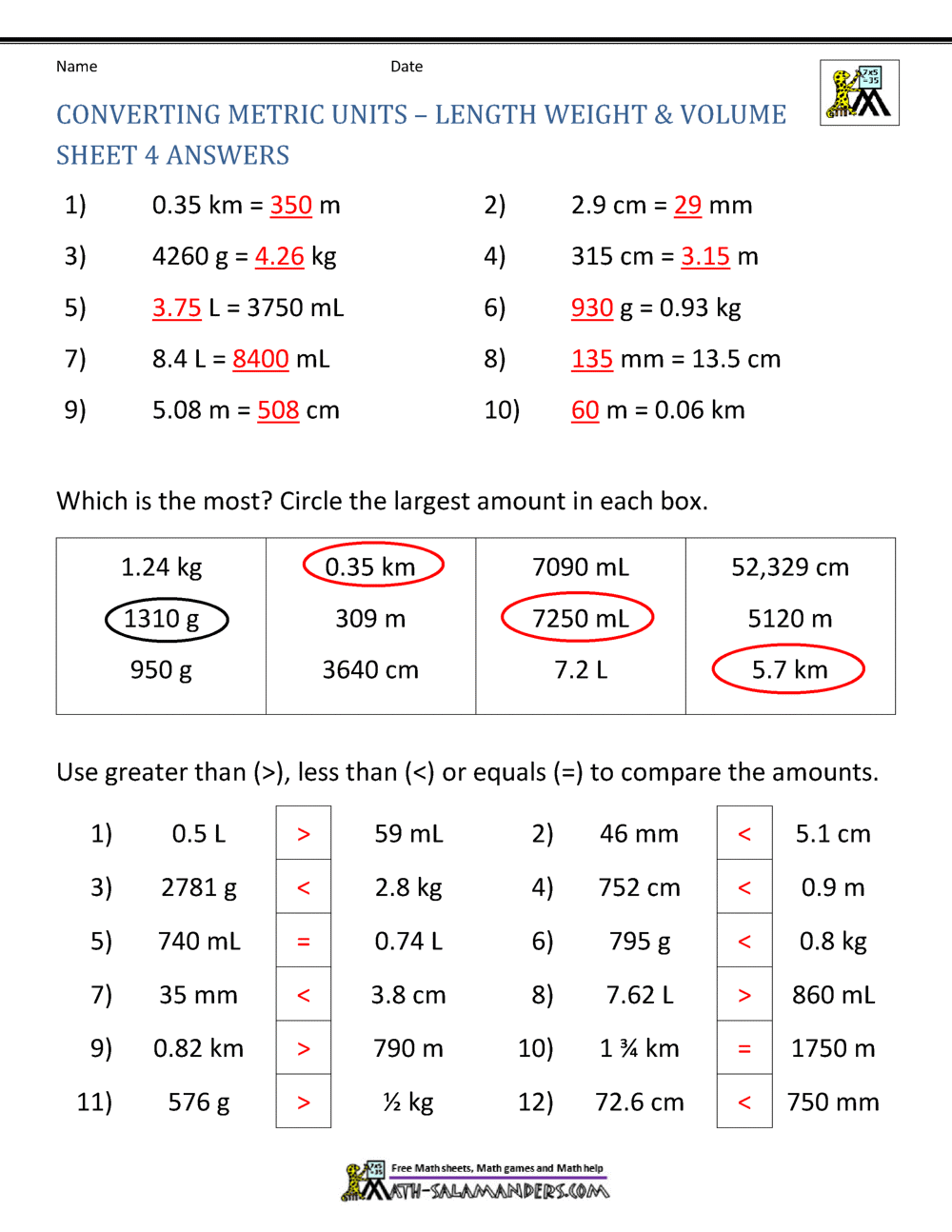
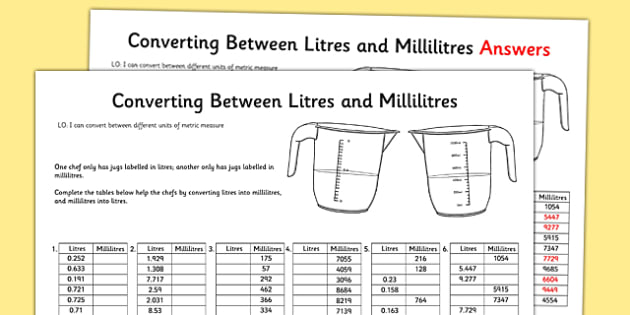
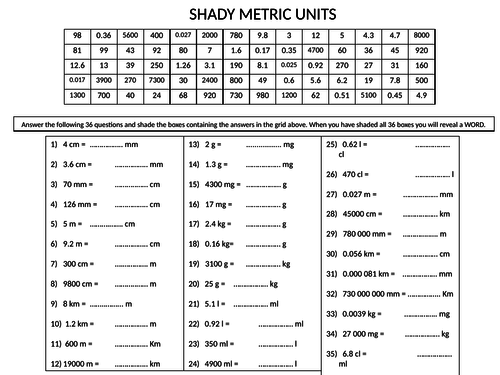
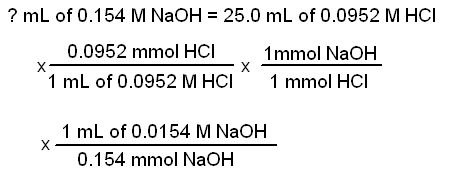What volume of water would you add to 15.00 mL of a 6.77 M solution of nitric acid in order to get a 1.50 M solution? | Socratic

2014 Mercedes-Benz M-Class ML 550 Stock # B1137C-DG for sale near Chicago, IL | IL Mercedes-Benz Dealer
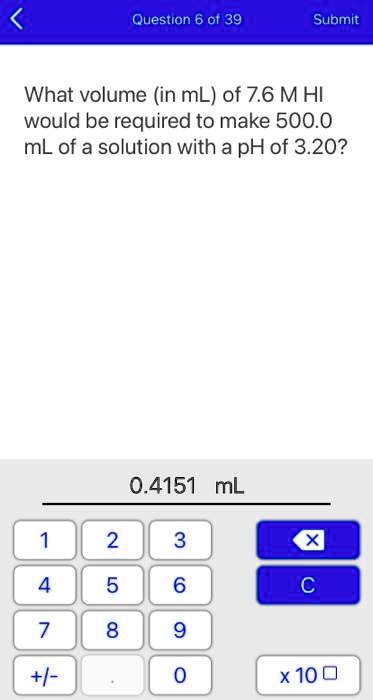
SOLVED:Question 6 of 39 Submit What volume (in mL) of 7.6 M HI would be required to make 500.0 mL of a solution with a pH of 3.20? 0.4151 mL 2 8 x10

Calculating concentrations. Different ways to express concentration: Number of grams of solute per liter of solution (g/L) Number of grams of solute per. - ppt download